In addition to encouraging the application of disease progression models to pharmaceutical research and development, the FDA has developed and made publicly available models for diseases of public health importance, including Alzheimer’s disease, Parkinson’s disease, bipolar disorder, obesity, and nonsmall cell lung cancer. Since that time, disease modeling has been widely adopted by the pharmaceutical industry. In 2004, the FDA launched its Critical Path Initiative, which aims to modernize drug development, and disease modeling was promoted as a key opportunity for advancement. Seminal work in disease progression modeling was published in the early 1990s, but the field has truly flourished in more recent years, since the US Food and Drug Administration (FDA) embraced such quantitative models as powerful tools that can be used to improve drug development productivity. In clinical pharmacology, disease models are often integrated with pharmacokinetic–pharmacodynamic models in order to quantify the influence of drug treatment on disease progression. Disease models may incorporate biomarkers of disease severity and/or clinical outcomes to characterize the natural progression of disease.

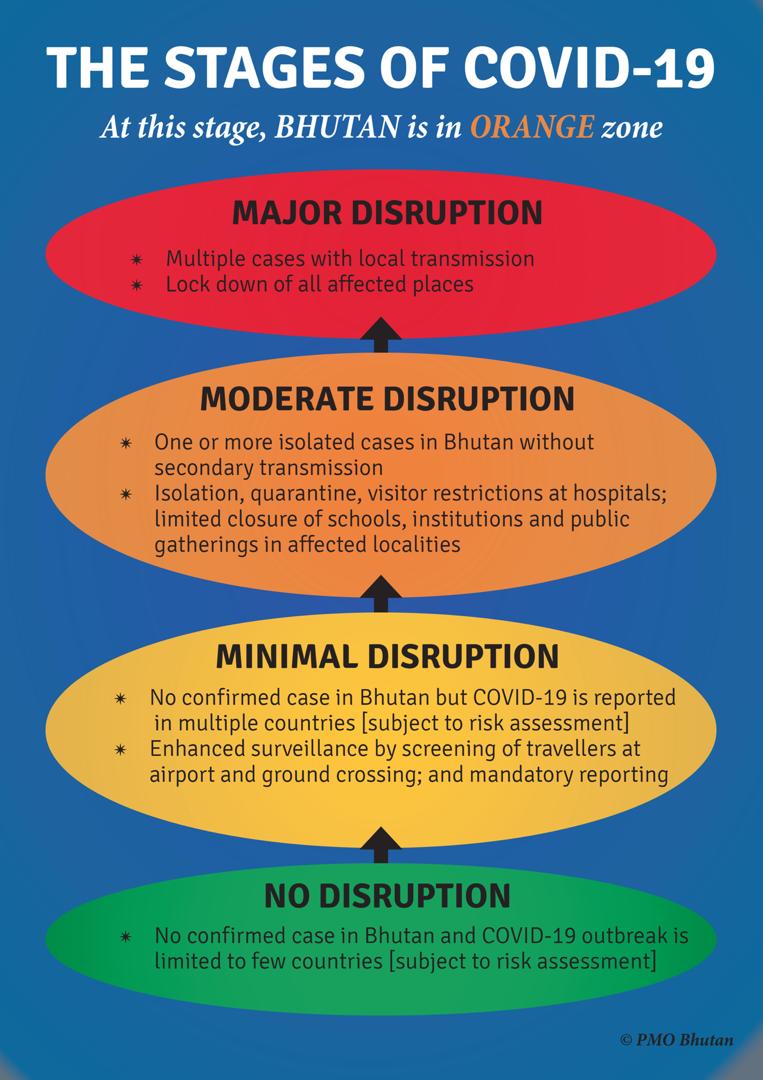
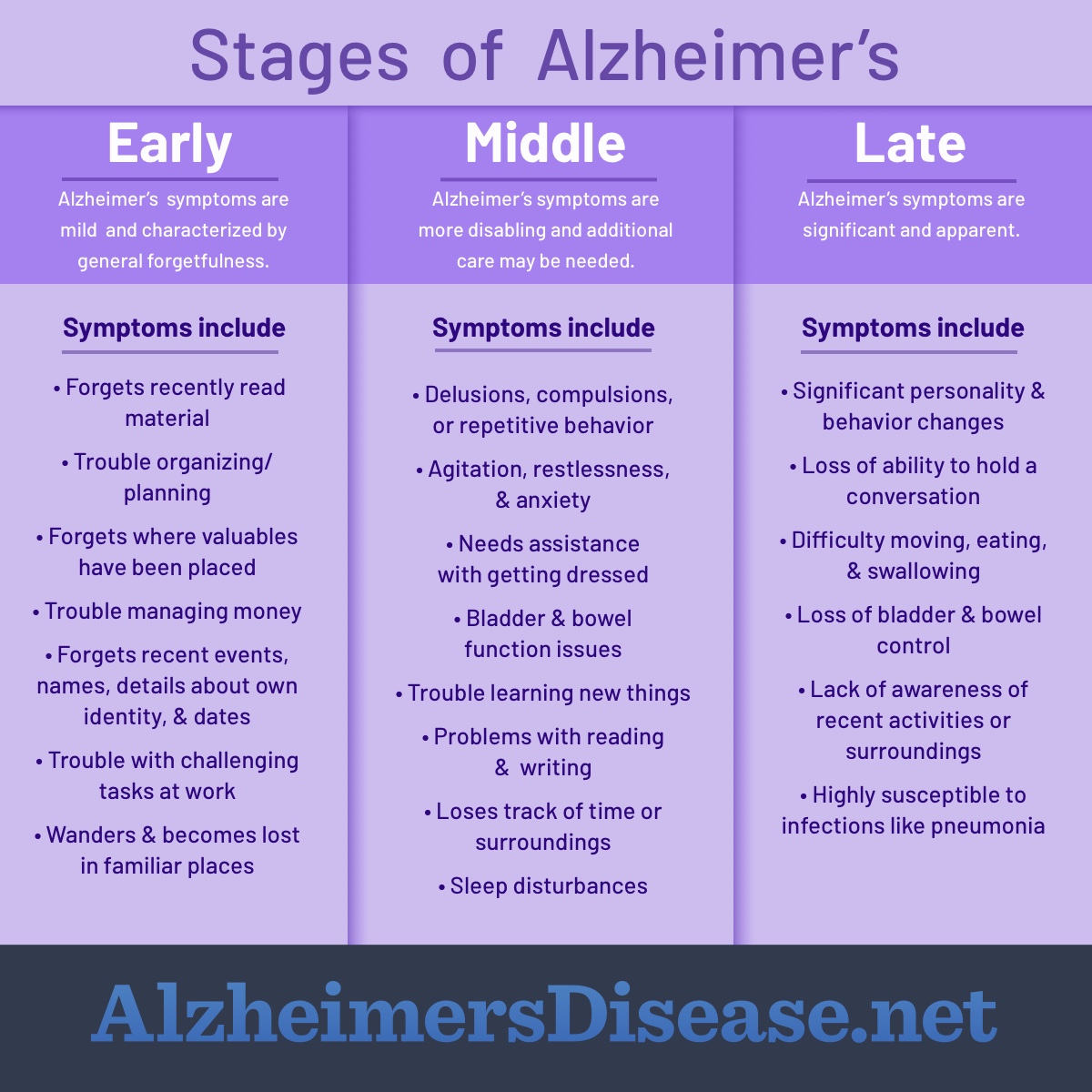
Disease progression modeling involves the utilization of mathematical functions as quantitative descriptors of the time course of disease status.
